|
The discovery of induced fission wouldn't have been possible without the strides made by Ernest Rutherford and Niels Bohr toward a coherent picture of the atom during the 1910s. The detonation of so-called 'atom bombs' is signified by the sight of a mushroom cloud - a dreadful reminder of the power of the atom and of fission itself. 2 Archived at the Wayback Machine.When this process is allowed to run unchecked, however, it gives rise to a powerful and destructive force. DOE Fundamentals handbook: Nuclear Physics and Reactor theory Vol.^ "RORSAT (Radar Ocean Reconnaissance Satellite)"."SNAP Overview – radium-219 – general background" (PDF). New York (NY): Reinhold Book Corporation. The Encyclopedia of the Chemical Elements. ^ a b Nuclear fission and fusion, and neutron interactions, National Physical Laboratory Archive.Total energy converted into heat in an operating thermal nuclear reactor Most modern nuclear weapon designs use plutonium-239 as the fissile component of the primary stage however, HEU (highly enriched uranium, in this case uranium that is 20% or more 235U) is frequently used in the secondary stage as an ignitor for the fusion fuel.Įnergy released when those prompt neutrons which do not (re)produce fission are captured Use of a large tamper, implosion geometries, trigger tubes, polonium triggers, tritium enhancement, and neutron reflectors can enable a more compact, economical weapon using one-fourth or less of the nominal critical mass, though this would likely only be possible in a country that already had extensive experience in engineering nuclear weapons. 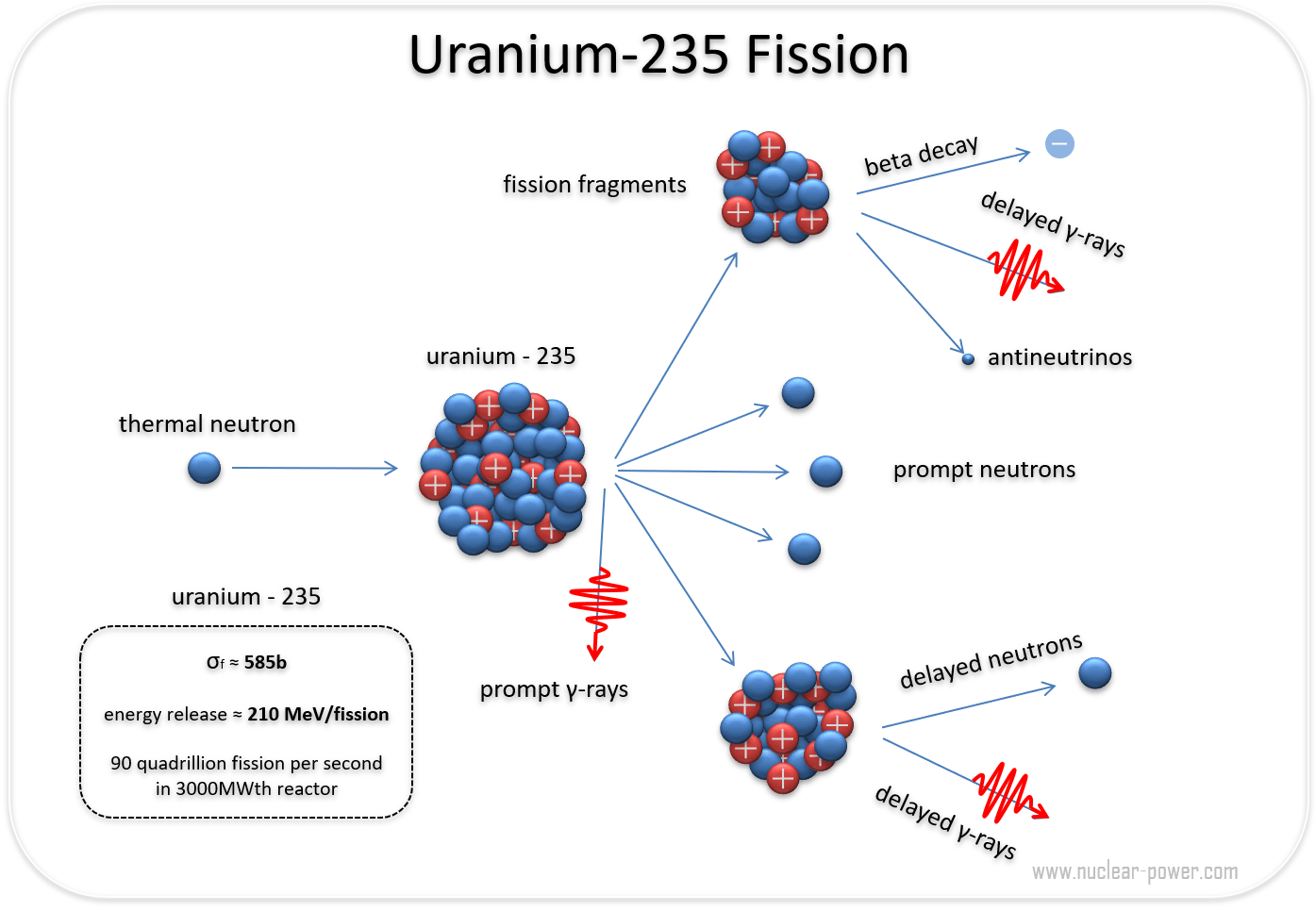
Even lower enrichment can be used, but this results in the required critical mass rapidly increasing. The material must be 85% or more of 235U and is known as weapons grade uranium, though for a crude and inefficient weapon 20% enrichment is sufficient (called weapon(s)-usable). The nominal spherical critical mass for an untampered 235U nuclear weapon is 56 kilograms (123 lb), which would form a sphere 17.32 centimetres (6.82 in) in diameter. The Little Boy gun-type atomic bomb dropped on Hiroshima on August 6, 1945, was made of highly enriched uranium with a large tamper. In nuclear bombs, the reaction is uncontrolled and the large amount of energy released creates a nuclear explosion. The power output of nuclear reactors is adjusted by the location of control rods containing elements that strongly absorb neutrons, e.g., boron, cadmium, or hafnium, in the reactor core. Some of them produce neutrons, called delayed neutrons, which contribute to the fission chain reaction. A fission chain reaction produces intermediate mass fragments which are highly radioactive and produce further energy by their radioactive decay. A critical chain reaction can be achieved at low concentrations of 235U if the neutrons from fission are moderated to lower their speed, since the probability for fission with slow neutrons is greater. If the reaction continues to sustain itself, it is said to be critical, and the mass of 235U required to produce the critical condition is said to be a critical mass. If at least one neutron from uranium-235 fission strikes another nucleus and causes it to fission, then the chain reaction will continue. Highly enriched uranium (HEU), which contains an even greater proportion of uranium-235, is sometimes used in the reactors of nuclear submarines, research reactors and nuclear weapons. Uranium enrichment removes some of the uranium-238 and increases the proportion of uranium-235. Heavy water reactors and some graphite moderated reactors can use natural uranium, but light water reactors must use low enriched uranium because of the higher neutron absorption of light water. Most neutron absorptions induce fission, though a minority result in the formation of uranium-236. For fast neutrons it is on the order of 1 barn. Its fission cross section for slow thermal neutrons is about 584.3☑ barns. It was discovered in 1935 by Arthur Jeffrey Dempster. 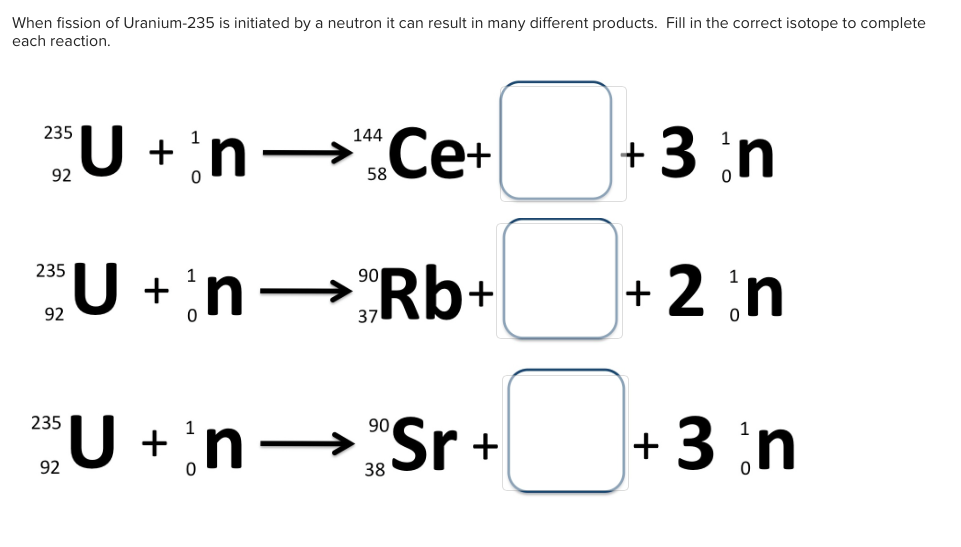
Uranium-235 has a half-life of 703.8 million years. It is the only fissile isotope that exists in nature as a primordial nuclide.  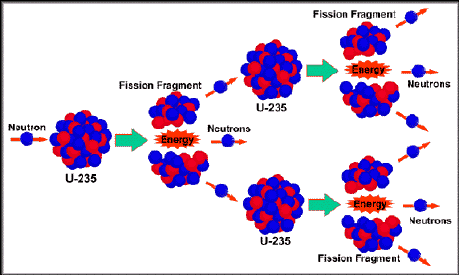
Unlike the predominant isotope uranium-238, it is fissile, i.e., it can sustain a nuclear chain reaction. Uranium-235 ( 235U or U-235) is an isotope of uranium making up about 0.72% of natural uranium. Uranium metal highly enriched in uranium-235
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
